RUNX1 is a master-transcriptional regulator involved in normal and malignant hematopoiesis. Germline, mono-allelic, missense mutations in RUNX1 cause large deletions or truncations that are mostly loss-of-function mutations. They cause familial platelet disorder (RUNX1-FPD), which evolves into myeloid malignancy (FPD-MM), e.g., MDS or AML. In addition to mutant (mt) RUNX1, FPD-MM cells commonly harbor co-mutations involving the second allele of RUNX1, or BCOR, PHF6, K-RAS, WT1 or TET2. Although curative in some patients, allogeneic stem cell transplantation from matched, un-related donors carries the risk of GVHD and relapse in FPD-MM. Thus, there is a need to develop novel targeted therapies to revert FPD-MM back to FPD.
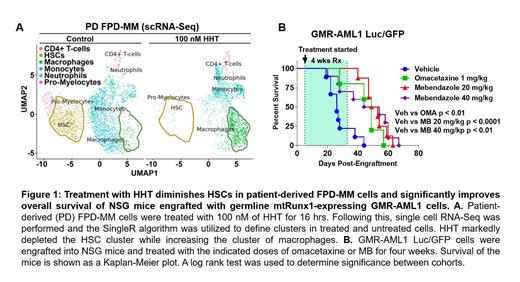
LINCS1000-CMap analysis, conducted with the RNA-Seq signature induced by lethal RUNX1 knockdown in AML cells with mutant (mt) RUNX1, was reported by us to reveal homoharringtonine (HHT or omacetaxine) and the anthelmintic fenbendazole (analog of mebendazole) as the top expression mimickers (EMs). In present studies, we demonstrate that treatment with HHT or mebendazole (MB) dose-dependently induced significantly greater loss of viability in six patient-derived (PD) samples of FPD-MM vs RUNX1-FPD or normal CD34+ progenitor cells. ATAC-Seq and H3K27Ac ChIP-Seq analyses conducted on BMA cells harvested longitudinally from the same patient at the RUNX1-FPD versus FPD-MM stage demonstrated greater ATAC-Seq peaks and chromatin accessibility in FPD-MM cells with increased mRNA expressions of MYB, MECOM and BCL2. Following HHT treatment, scRNA-Seq analysis demonstrated a marked reduction in the population of HSCs but concomitant increase in the macrophage population in FPD-MM (Fig. 1A). This was associated with positive enrichment of the mRNA expression belonging to the gene-sets (HALLMARK) of apoptosis signaling and TP53 targets while negatively enriching those of c-Myc targets. CyTOF analysis demonstrated that HHT treatment reduced c-Myc, EVI1, MCL1, BFL1 and CDK6 expression in phenotypically characterized FPD-MM stem-progenitor cells.
Utilizing an FPD-MM sample from a patient with Runx1 K194N, we established the first ever cell line (GMR-AML1) expressing germline mtRUNX1. GMR-AML1 cells exhibited high surface expression of CD117 (c-KIT), CD123 (IL-3R) and CD33. CRISPR knockout (KO) RUNX1 led to growth inhibition, such that GMR-AML1 cells that retain Runx1 outcompeted Runx1 KO cells within 20 days. Treatment with HHT induced loss of viability in GMR-AML1 cells (LD50: 40 nM). HHT also caused concordant decline in ATAC-Seq and RNA-Seq peaks in ribosomal genes involved in protein translation, DNA POLR2B, RRM2, CCND2 and PTPN11. Treatment with MB also dose-dependently induced loss of viability of GMR-AML1 cells (LD50: 330 nM) as well as inhibited cell cycle and protein translation (via OPP Assay). CyTOF analysis revealed that MB treatment also depleted CDK6, RUNX1, c-Myc and EVI1 in phenotypically defined FPD-MM stem cells. Finally, in the tail-vein infused and engrafted, luciferized GMR-AML1 cell xenograft, compared to the vehicle control, treatment with omacetaxine or MB significantly reduced AML burden and improved overall survival of the NSG mice (p < 0.05) (Fig. 1B). These preclinical findings highlight the molecular features associated with progression of RUNX1-FPD to FPD-MM, including the newly established GMR-AML1 cell line. They also demonstrate that HHT or MB are effective against cellular models of FPD-MM versus RUNX1-FPD.
Disclosures
DiNardo:Notable Labs: Honoraria; ImmuniOnc: Honoraria; AbbVie/Genentech: Honoraria; Fogham: Honoraria; Servier: Honoraria; Novartis: Honoraria; Takeda: Honoraria; Schrödinger: Consultancy; Astellas: Honoraria; BMS: Honoraria. Loghavi:Caris Diagnostics: Consultancy; Blueprint Medicine: Consultancy; Abbvie: Consultancy; Gerson Lehrman Group: Consultancy; QualWorld: Consultancy; Guidepoint: Consultancy; Recordati/ EUSA Pharma: Consultancy; Daiichi Sankyo: Consultancy; Astellas: Research Funding; Amgen: Research Funding; Abbvie: Current equity holder in publicly-traded company. Bhalla:Foghorn Therapeutics Inc.: Research Funding.